Home / Health / UK Approves Groundbreaking Alopecia Treatment
UK Approves Groundbreaking Alopecia Treatment
12 Mar
Summary
- A new pill, deuruxolitinib, is approved for severe alopecia areata.
- Nearly a third of patients regrew 80% of scalp hair in trials.
- The treatment works by reducing immune system attacks on hair follicles.
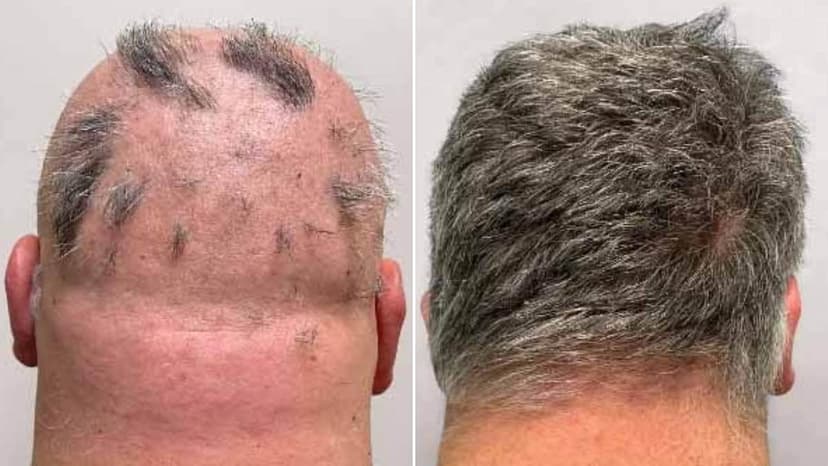
In Britain, a new treatment for severe alopecia areata has received approval from the MHRA. Deuruxolitinib, marketed as Leqselvi, offers a promising option for adults suffering from significant hair loss.
Clinical trials demonstrated remarkable results, with nearly 30% of participants regrowing at least 80% of their scalp hair within a six-month period. An additional 25% achieved regrowth of 90% or more, providing substantial hope for those affected by this distressing autoimmune condition.
The mechanism of action for deuruxolitinib involves reducing inflammation and preventing the immune system from mistakenly targeting hair follicles. This allows dormant follicles to reactivate and promote hair regrowth, a significant development for alopecia sufferers.
While the drug is now available via private prescription, its availability through the NHS hinges on an assessment by NICE for value for money. Common side effects reported in trials included headaches and acne, affecting over 10% of patients.
This approval follows the 2024 licensing of Ritlecitinib, another Janus kinase (JAK) inhibitor, for severe alopecia areata in adolescents and adults. Both treatments represent advances in managing the condition, which affects approximately one in 500 people.




