Home / Health / Japan Approves World's First iPS Cell Therapies
Japan Approves World's First iPS Cell Therapies
6 Mar
Summary
- Japan approved new stem-cell treatments for Parkinson's and heart failure.
- These therapies are the first commercial products using iPS cells.
- Treatments could be available to patients as early as summer.
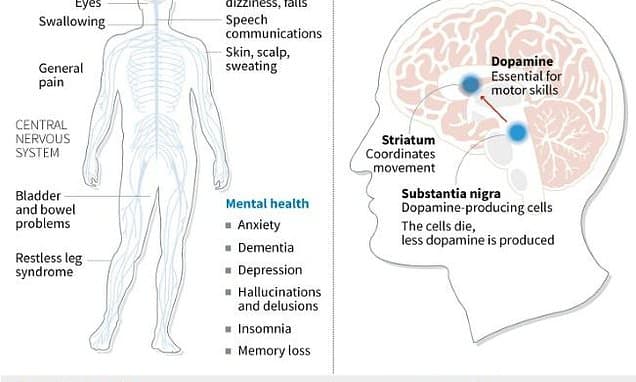
Japan has achieved a major medical milestone by approving groundbreaking stem-cell therapies for Parkinson's disease and severe heart failure. These treatments, developed using iPS cells, are poised to become the world's first commercially available medical products of their kind. Sumitomo Pharma's "Amchepry" has received conditional approval for Parkinson's, involving the transplantation of iPS cells into the brain to replace dopamine-producing cells. Early trial data indicated safety and symptom improvement in some patients. Cuorips' "ReHeart" heart muscle sheets, designed to restore heart function by forming new blood vessels, also secured approval. These revolutionary therapies are anticipated to reach patients as early as this summer, offering new hope to millions affected by these debilitating conditions. The approval system is designed to expedite patient access to such innovative treatments.