Home / Health / FDA Warns Parkinson's Treatment May Cause Seizures
FDA Warns Parkinson's Treatment May Cause Seizures
20 Mar
Summary
- FDA requires new seizure risk warning on Parkinson's drugs.
- Carbidopa/levodopa treatments may cause vitamin B6 deficiency.
- 14 seizure cases linked to vitamin B6 deficiency reported.

The U.S. Food and Drug Administration has issued a notification to manufacturers of certain Parkinson's disease medications, requiring updated prescribing information. This update mandates a new warning concerning a potential risk of seizures associated with these treatments.
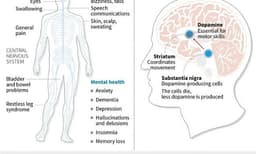
The health regulator's safety review revealed 14 instances of seizures linked to vitamin B6 deficiency in patients using drugs containing carbidopa or levodopa. These medications, typically administered together to manage Parkinson's symptoms, can deplete vitamin B6 levels during their mechanism of action.
Healthcare professionals are advised to assess vitamin B6 levels in patients before initiating these Parkinson's treatments and consider supplementation. The FDA's updated guidance aims to ensure patient safety by highlighting this potential adverse effect.