Home / Health / Blood Pressure Meds Switched: Urgent Recall Issued
Blood Pressure Meds Switched: Urgent Recall Issued
6 Mar
Summary
- Ramipril 5mg Capsules batch GR164099 recalled due to packaging error.
- Packs may contain Amlodipine 5mg Tablets instead of Ramipril.
- Patients with affected packs should return them to their pharmacist.
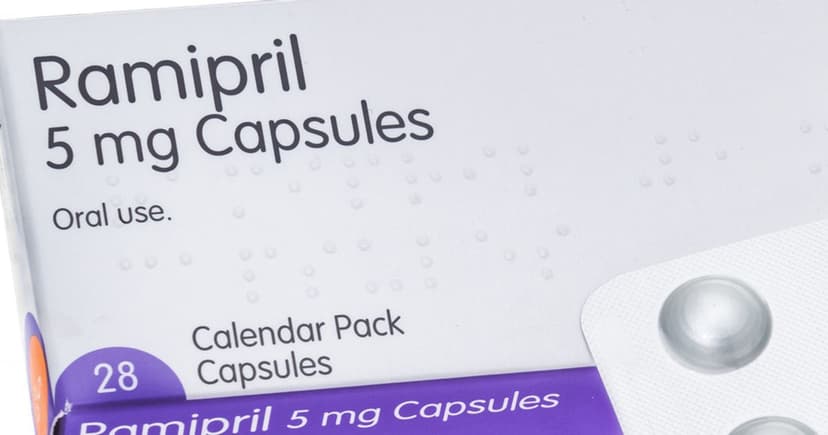
Crescent Pharma Limited is conducting a precautionary recall of Ramipril 5mg Capsules, batch GR164099. This action follows a complaint where a patient reported receiving blister strips of Amlodipine 5mg Tablets within a box labelled as Ramipril 5mg Capsules. The error is believed to have occurred during the packaging process at a facility manufacturing both blood pressure medications. Patients prescribed Ramipril are urged to verify their packaging for the specific batch number and to return any packs containing Amlodipine blister strips to their pharmacy.
The Medicines and Healthcare products Regulatory Agency (MHRA) states the likelihood of patients receiving the wrong medication is low. However, if Amlodipine 5mg Tablets were inadvertently taken, individuals might experience dizziness due to potentially lower blood pressure. The MHRA advises those experiencing side effects to seek immediate medical advice and to bring their medication and any accompanying leaflet to their pharmacy or GP. Healthcare professionals have been instructed to return all remaining affected stock to their suppliers.