Home / Health / HIV Drug Access Expanded to Millions
HIV Drug Access Expanded to Millions
15 Apr
Summary
- Goal to reach 3 million people with HIV prevention drug by 2028.
- Lenacapavir, a twice-yearly injection, aids HIV prevention.
- Generic manufacturers will produce more affordable versions.
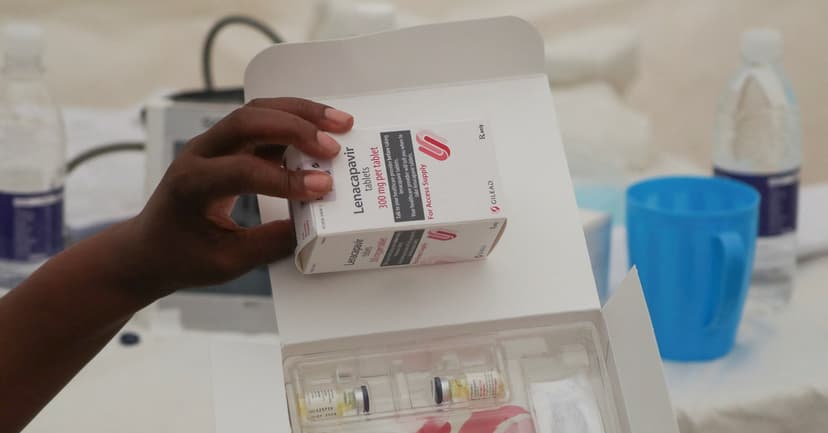
The U.S. and the Global Fund to Fight AIDS, Tuberculosis and Malaria are intensifying efforts to broaden access to Gilead's HIV prevention drug, lenacapavir. Their ambitious goal is to reach 3 million people by the year 2028.
Initial shipments of lenacapavir, a subcutaneous injection administered twice annually, have successfully reached nine African countries. The Global Fund announced its intention to extend the drug's provision to twelve additional countries, including the Dominican Republic, Fiji, Indonesia, Morocco, Rwanda, and Thailand.
This expanded access follows a prior agreement in July of last year between the Global Fund and Gilead to supply lenacapavir to low-income nations, with the aim of reaching up to 2 million people over three years.
Lenacapavir's twice-yearly injection format offers an advantage over daily oral pre-exposure prophylaxis (PrEP) pills, addressing issues like missed doses and supply chain inconsistencies. Early data highlights strong uptake among key groups, such as pregnant and breastfeeding women, adolescent girls, and young women, as well as individuals new to such preventive treatments.
To ensure widespread and affordable availability, Gilead has granted voluntary licenses to multiple manufacturers for the production of generic lenacapavir. This strategy is expected to facilitate expanded and more cost-effective supply in the future, as stated by the Global Fund. The organization is actively working to ensure sustainable and affordable access at scale by diversifying its supplier base.