Home / Health / FDA Expands Libido Pill Access Post-Menopause
FDA Expands Libido Pill Access Post-Menopause
16 Dec, 2025
Summary
- FDA broadens Addyi's use to women over 65 post-menopause.
- Addyi carries a warning for dangerous interactions with alcohol.
- Low libido drug development surged after Viagra's success.
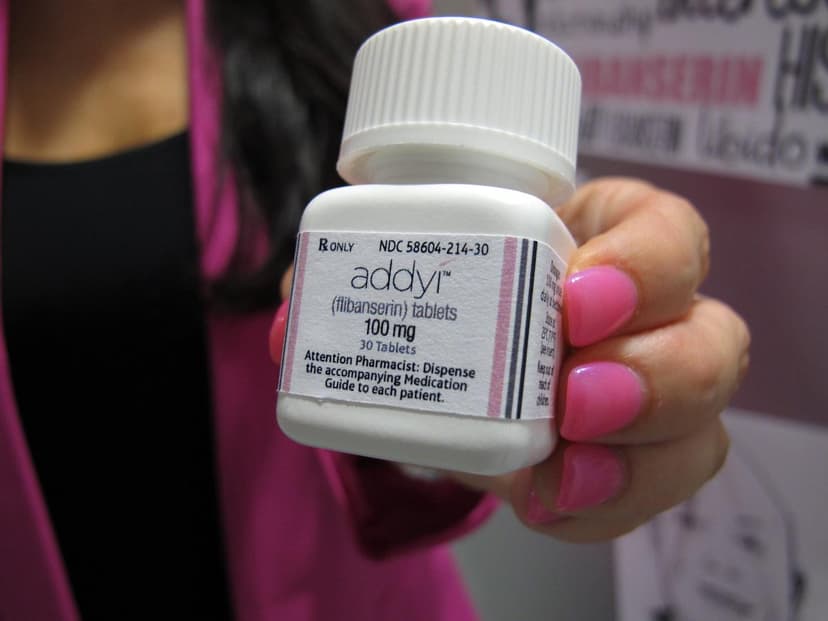
U.S. health officials have expanded the approval of Addyi, a medication designed to boost libido in women. The Food and Drug Administration announced that the once-daily pill can now be prescribed to women over the age of 65 who have completed menopause. This decision broadens the accessibility of the drug, which was first approved a decade ago for premenopausal women experiencing emotional distress due to low sex drive.
Addyi, marketed by Sprout Pharmaceuticals, was initially anticipated to be a major success in women's health. However, its limited sales can be attributed to notable side effects like dizziness and nausea. Critically, the drug carries a serious safety warning regarding the consumption of alcohol, which can lead to dangerously low blood pressure and fainting. This cautionary note underscores the complex safety profile of the medication.




