Home / Health / MHRA Warns of Falsified Mounjaro Pens: Check Batch Numbers!
MHRA Warns of Falsified Mounjaro Pens: Check Batch Numbers!
27 Feb
Summary
- Counterfeit Mounjaro pens discovered, posing infection risk.
- Five affected pens were distributed via a Birmingham online pharmacy.
- Pens appear to contain tirzepatide but sterility is unconfirmed.
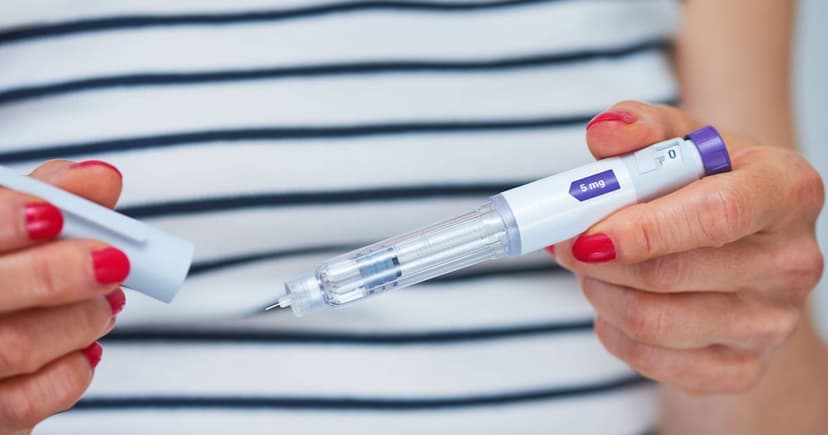
The Medicines and Healthcare products Regulatory Agency (MHRA) has issued a safety warning concerning falsified Mounjaro pens distributed to the public. An online pharmacy was identified as supplying pens that did not meet safety standards. Five of these affected Mounjaro KwikPen 7.5mg pens were supplied through the Private Pharmacy Clinic in Birmingham.
Testing confirmed the presence of tirzepatide, the active substance in Mounjaro. However, the manufacturing process is unknown, raising concerns about sterility. This lack of sterile preparation presents a risk of infection or allergic reactions for users. Symptoms of infection can include redness or warmth at the injection site, while allergic reactions may involve rashes or breathing difficulties.
So far, no patients have required treatment due to these counterfeit medications. The issue came to light after the genuine manufacturer, Eli Lilly, reported faulty pens. Users are urged to check their pen's batch number and strength. If an affected pen is found, it should be stopped immediately and reported to the MHRA. Healthcare professionals are available for guidance.