Home / Business and Economy / India's Generics Flood: Ozempic, Wegovy Prices Slashed
India's Generics Flood: Ozempic, Wegovy Prices Slashed
20 Mar
Summary
- Over 40 Indian drugmakers will launch generics at half the price.
- Semaglutide ingredient patent expiry expected to halve drug costs.
- Concerns rise over regulatory oversight and potential drug misuse.
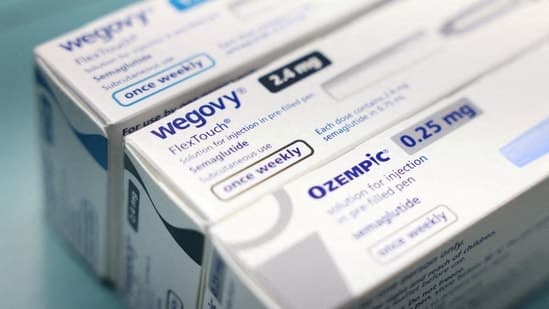
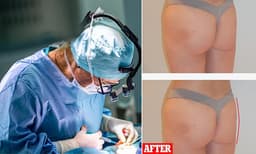
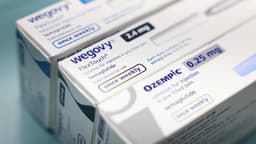
As of today, March 20, 2026, India's pharmaceutical sector is poised to dramatically increase access to weight-loss medications. Following the expiration of a crucial patent for the semaglutide ingredient, over 40 Indian drugmakers are preparing to launch more than 50 brands. These generics are anticipated to be priced at roughly half the cost of existing blockbuster drugs like Ozempic and Wegovy.
The market is projected to surge from approximately ₹1,500 crore to ₹8,000 crore by 2030. Monthly costs for semaglutide treatments are expected to decrease from around ₹11,000 to ₹3,000-₹5,000 almost immediately. This sharp price reduction could enable patients from lower economic strata to access these treatments, though maintaining pricing discipline will be key for corporate profitability.
Strategic collaborations are emerging, such as the licensing agreement between Zydus Lifesciences and Torrent Pharmaceuticals for a semaglutide injection. Zydus will manufacture an innovative formulation, while Torrent Pharma will market it under the brand name Sembolic. This partnership allows Zydus to leverage its manufacturing capabilities and Torrent to utilize its extensive field force.
However, the influx of affordable generics is also sparking concerns about regulatory oversight. Experts warn of potential issues such as direct pharmacy purchases, leakages through distributors, or non-medical use, particularly in urban areas. Such unregulated access could lead to poor dosage management and adverse effects, potentially prompting stricter regulatory measures from the government.
In the long term, physician trust and reliable supply chain integrity will be critical for success. Weaker players with quality issues are predicted to exit the market within two to three years, leaving established companies that guarantee consistent patient outcomes.